
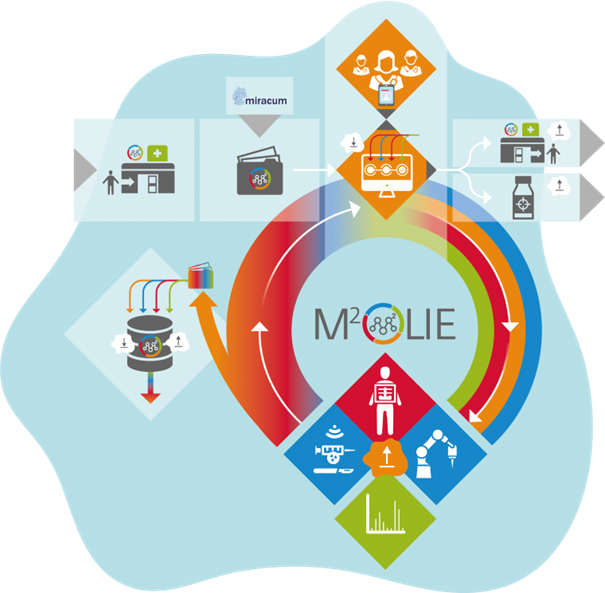
The goal of M²OLIE in the third and final funding phase is to complete the basic process established in the second funding phase with the aim of creating a comprehensive M²OLIE closed-loop process for the diagnosis and treatment of patients with oligometastases within a limited period of time. To ensure the holistic design of this process, four modules were introduced in the third funding phase, which can be flexibly combined in the future for different procedures and applications in other indications.
The process begins with patient admission, where the TEDIAS system will be integrated into the closed-loop process in the future. The fully digital and comprehensive process management system, the M²OLIE cockpit, guides the physicians through the various process steps.
The Operational Platform module is designed to connect all subcomponents of the M²OLIE closed-loop process to the cockpit and transfer them into the real clinical process.

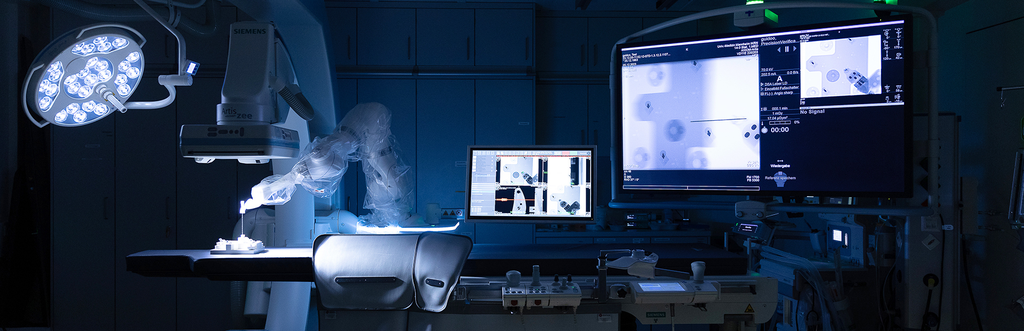
The Interventional Platform module creates an intervention environment that significantly increases efficiency and precision at the diagnostic, therapeutic, and organizational levels through a higher degree of digitization and automation. When analyzing the biopsy, the Laboratory module will focus on the diagnostic and therapeutic addressing of tumor heterogeneity in order to achieve a complete, highly efficient, and partially automated characterization of tumor foci. The Tumor Board module aims to establish an ad hoc and asynchronous tumor board that can be convened and conducted using a mobile app and in which the guidelines are already integrated.
The establishment of the processes forms the basis for transferring the results to the planned M²OLIE clinic as an important pillar of the continuation strategy. In addition, the declared goal of all M²OLIE partners involved is to transfer the entire process or individual modules to other clinics so that tumor patients elsewhere can also benefit from this revolutionary diagnosis and treatment process.
The Laboratory module develops approaches to achieve comprehensive, highly efficient, AI-supported, and partially automated characterization of tumor biopsies and resections. The aim is to characterize and quantify the heterogeneity of the disease so that subsequent therapy can be optimized and tailored to the individual lesion. This includes the acceleration, completion, and systematic examination of all potentially clinically relevant tissue parameters from biopsies and resections, as well as the automated collection, evaluation, and digitization of data as additional information for the tumor board by means of a complete pathological examination of human tissue, including specialist medical findings and complete digitization of the data. This is supplemented by state-of-the-art methods of multiplex immunohistochemistry mass spectrometry imaging for the fine tissue characterization of malignant lesions and their evaluation and validation for routine clinical use in the context of a clinical study.
Mass spectrometry enables rapid tissue classification within interventional radiology and tumor (sub)classification for targeted therapy decisions. Optimally adapted antibodies are also being developed for this purpose to cover the clinically relevant spectrum of applications. On the other hand, conventional pathological diagnosis is further supplemented by novel methods of tissue preparation, optical imaging, and automated AI-based image analysis to obtain representative insights into the density distribution of tumor cells. For this purpose, the samples are examined as a whole in order to investigate cell stratification and the distribution of relevant parameters within the lesion and to develop a solid database on how many tissue sections are required for a solid pathological diagnosis. In addition, the laboratory module also researches tumor-specific nuclear medicine diagnostics and therapy options as well as their automation, which are available for endoradiotherapy of the lesions if they match the profile of the tumor lesions.
In the routine clinical context of pathological diagnosis, human tissue taken from the patient is characterized using standard procedures in accordance with guidelines within one week, the findings are compiled, and then made available to the tumor board. The analyses include individual, selected parameters to be examined, the relevance of which has already been proven in studies. Within the framework of the M²OLIE closed-loop process, however, the specialist findings should be available within hours of sampling, and the analysis and evaluation should include as many potentially relevant parameters as possible, allowing for a better assessment of the disease/individual lesion in order to make an optimally tailored therapy decision for the individual patient.The scientific and technical objectives therefore include, on the one hand, the highly efficient and partially automated characterization of the biopsies obtained, including parameters that go well beyond those usually examined, with the aim of comprehensively and thus completely characterizing the individual lesions. As in other areas of the research campus, this aims to address tumor heterogeneity, which is the basis for controlling oligometastatic tumor disease. Likewise, the automated collection, evaluation, and digitization of data is being developed as additional information for the tumor board by means of complete pathological examination of human tissue, including specialist medical evaluation and digitization of the data.
To support pathological diagnosis, the extent of tumor heterogeneity in M²OLIE-relevant lesions is evaluated. To this end, novel methods of tissue preparation, optical imaging, and automated AI-based image analysis are being developed and used to obtain representative insights into the density distribution of tumor cells. It is crucial that the tissue samples remain intact as a whole so that cell stratification and the distribution of tumor foci with high division activity become visible. Among other things, this allows us to determine the minimum number of regular tissue sections required for a solid pathological diagnosis. This increases diagnostic reliability and, where appropriate, reduces the amount of work and time associated with conventional diagnosis while maintaining the same diagnostic quality.
In the field of molecular medicine, imaging and mass spectrometry techniques are being used in collaboration with our corporate partner PROGEN Biotechnik to research the use of multiplex immunohistochemistry mass spectrometry for the tissue characterization of malignant lesions and to evaluate and validate its routine clinical use in a clinical study. This has two implications for the M²OLIE closed-loop process. On the one hand, a targeted therapy decision can be made on the basis of tumor (sub)classification to improve patient care. For this purpose, multiplex immunohistochemistry mass spectrometry is used to simultaneously read out a large number of biomarkers on a single tissue section after biopsy, provided that suitable antibodies are available, and to analyze and interpret their co-localization in the tissue. On the other hand, the approach can be used as a diagnostic tool in molecular medicine to enable rapid tissue classification within interventional radiology. This serves to characterize the targets of theranostic ligands, which could subsequently be used as targeted, tumor-specific therapeutics. This means that in the future, the M²OLIE Clinic will have one of the most modern biomedical analysis methods for tissue diagnostics at its disposal.
Currently, there are insufficient numbers of optimally adapted tumor-specific radiopharmaceuticals available for the sensitive and specific imaging of target lesions using PET and their highly efficient targeted therapy using endoradiotherapeutics. Ideally, these theranostic radiopharmaceuticals should also be able to address and overcome the heterogeneity of tumor disease. As part of this work, new radiopharmaceuticals for the complete and highly sensitive imaging and characterization of tumor foci are therefore being researched, and the findings are being applied to the development of endoradiotherapeutics adapted to the disease (in particular α-emitters based on antibodies, their fragments, and nanoparticulate systems) adapted to the disease, and, in collaboration with the company partner Elysia, their GMP-compliant automated production for clinical application is being researched.
In the closed-loop process, the Operational Platform module performs three key functions: It acts as a central control element that links and coordinates all participating modules on the IT side. In addition, it is involved in the diagnosis process, where it makes a significant contribution to improving quality and efficiency in medical decision-making. The module also complements the project by focusing on the patient, mapping the closed-loop process from the patient’s perspective through a fully integrated mobile patient application and digitally guiding and supporting patients through each step of the process.
As the technical backbone, the platform controls all process steps via the M²OLIE cockpit – from admission and medical history to the planning of diagnostic and therapeutic measures. At the same time, it serves as a central data platform that supplies all participating modules and is integrated into the IT infrastructure of the University Medical Center Mannheim. Structured and standardized communication between the modules is ensured, for example, through the use of FHIR (Fast Healthcare Interoperability Resources) interfaces. This enables seamless, interoperable, and secure data transfer within the system and with external clinical IT systems—an essential factor for an efficient closed-loop process.
Another key element is the integration of innovative algorithms for image processing and interpretation. These enable automated and standardized analysis of radiological image data, thereby optimizing the diagnostic process. On this basis, precise 3D biopsy planning is possible, allowing targeted and minimally invasive puncture of suspicious lesions. This increases both diagnostic accuracy and patient safety. Another important component is the application of radiomics: Here, extensive quantitative features are extracted from image data, which can provide additional information about tumor biology and response to therapy.
For patients with oligometastases, these integrations significantly improve the patient journey: the IT-supported treatment process enables structured, fast, and safe care. The continuous availability and optimized evaluation of image and patient data lead to efficient therapy decisions. Doctors and patients are guided continuously through the process—transparently, safely, and effectively.
The M²Clinic subproject aims to technically integrate the M²OLIE clinic into the IT infrastructure of the University Medical Center Mannheim (UMM), thereby laying the foundation for the complete M²OLIE closed-loop process. This process forms the core of the M²OLIE project and connects all clinical and technical modules along the patient journey—from admission to therapy—into a closed, digitally supported care pathway.
An important foundation was laid in the second funding phase with the connection of the first clinical information systems to the M²OLIE Data Lake. The third funding phase will build on this: the focus is now on connecting the intervention platform, laboratory, tumor board, and patient-centered systems such as a patient app and the TE-DIAS admission tool. MaLu-IT, the IT department of the UMM, plays a key role in this. It provides hardware, software, and IT expertise, conducts interface workshops, and trains researchers in clinical IT processes.
The aim is to ensure smooth integration through standardized interfaces (e.g., FHIR) and existing systems—without additional development on the part of the hospital. The ProM²etheus platform is linked to clinical workflows so that processes can run automatically and in a coordinated manner. Technically, this includes the use of powerful server architectures. M²Clinic thus creates the technical basis for clinical studies, research on real data, and the IT implementation of the M²OLIE clinic. It bundles the IT results of all modules and makes them usable in the overall process. Without this integration, the technical completion of the closed loop would not be feasible.
The M2OLIE-PAL subproject aims to expand the existing closed-loop process to include a patient-centered approach. This will be achieved by developing a mobile application that provides patients with the best possible support throughout the entire patient journey, from digital patient education and admission to diagnosis and therapy. The focus is on intuitive use and patient requirements.
The application not only supports the process, but also enables the measurement of objective (PROMs) and subjective (PREMs) quality indicators for a comprehensive evaluation of patient satisfaction. In addition, patients are supported in the clinical process through interactive communication with information on the course of treatment, navigation aids, and a digital patient file. Digital patient admission is partially automated and integrated using the TEDIAS booth from Fraunhofer IPA.
M2OLIE-PAL aims to ensure positive patient experiences through continuous support and satisfaction measurement. This is made possible by the use of existing communication protocols (HL7 FHIR), an integrative central data supply (data lake), and separate patient interfaces. This combines the previous treatment-centered approach with a patient-centered perspective, which promotes acceptance of the M²OLIE closed loop. The future use of AI technologies (e.g., generative language models for psychosocial support) will be researched in greater depth in order to further increase patient centricity and actively improve patient satisfaction.
The aim of this subproject is to complete the M²OLIE closed-loop process. Within the M²OLIE Operational Platform, approaches for further developing the M²OLIE cockpit into a demonstrator within the physician journey are being researched, and the implementation of 3D intervention planning incorporating image-based information, tumor characterization based on PCCT (photon-counting CT), and AI-supported evaluations, as well as incorporating existing risk structures. A correlation between pre-interventional imaging (CT/PCCT/MRI/PET) and biopsy information and a resulting adaptation of the intervention planning is also being researched.
The “Tumor Board” module addresses the central role of interdisciplinary tumor boards in the care of patients with oligometastases. The aim is to sustainably improve planning, implementation, and decision-making in the tumor board through structured, digital, and data-driven support, thereby increasing the quality and efficiency of treatment decisions. The module is located in the closed-loop process at the interface between diagnostics, therapy planning, and clinical decision-making. It integrates relevant image, patient, and diagnostic data from various sources, processes it in a structured manner, and makes it available to the tumor board in a context-sensitive manner. The decisions made in the tumor board are then fed back into downstream therapy and evaluation processes and form the basis for continuous learning and optimization.
In terms of content, the module builds on preliminary work carried out by the research campus, in particular on existing digital infrastructures, data models, and initial applications to support clinical decision-making processes. In the third funding phase, the focus is on the integration, expansion, and clinical validation of these approaches in everyday tumor board practice. For patients with oligometastases, this means an improved patient journey: relevant information is available in a complete and structured form, treatment options can be evaluated in an interdisciplinary manner based on guidelines, and decision-making processes become more transparent and comprehensible. This should reduce delays, make treatment decisions more precise, and improve the overall quality of care.
Collaboration between science and industry is a central element of the module. Clinical partners define requirements from everyday care and contribute medical expertise, while industrial partners develop and implement digital solutions and optimize them in terms of scalability, user-friendliness, and integration into existing systems. Development is iterative and application-oriented. Patients are indirectly involved in the research through the systematic consideration of clinical outcomes, the evaluation of care processes, and, in the future, patient-reported endpoints. The goal is the consistent, patient-centered further development of the tumor board process.
The aim of this sub-project is to provide interfaces for integrating clinical data into the c37.TumorBoard. This includes connecting the QIT PACS to the hospital PACS for the integration of diagnostic imaging. Basic clinical data on patients is transferred via patient data interfaces to the HIS. An external data interface also enables the integration of specialized findings and analyses from pathology, diagnostics, and genetics. In addition, a connection to Prometheus/Cockpit is established in order to integrate further relevant data sources into the overall process. The project partners are celsius37, Mannheim University Hospital, Heidelberg University, and MintMedical. In the M²OLIE process, the subproject is located after biopsy, laboratory, and diagnostics. It creates the technical and procedural basis for structured case discussion in the c37.TumorBoard.
The aim of this subproject is to create templates for treatment procedures and to prepare patient presentations for the tumor board in a structured manner. Templates for treatment procedures are being developed to support complete and quality-assured case preparation. In addition, preparations are to be made for the compact presentation in the tumor board in order to enable a clear and efficient presentation of all information relevant to decision-making. The cooperation partners in this project are celsius37, Mannheim University Hospital, and Heidelberg University. In the closed-loop process, the subproject is located between biopsy, laboratory, and diagnostics in the transition to therapy and supports structured therapy planning in the tumor board.
In this subproject, the tumor board process in c37.TumorBoard will be specifically optimized. This includes adapting the registration form with automatic and patient-specific data import of imaging, laboratory, and pathology results via the existing interfaces. The display of imaging within the tumor board is to be optimized, and direct access to all relevant findings is to be available during case discussions. In addition, an ad hoc or asynchronous tumor board application will be developed and integrated into the celsius37 system. This solution enables flexible, time- and location-independent processing of cases, especially via mobile devices such as tablets. This makes interdisciplinary coordination possible even outside of regular tumor board hours, which significantly increases efficiency and response speed in the clinical decision-making process. The project partners are celsius37, Mannheim University Hospital, and Heidelberg University. The subproject is part of the closed-loop process following biopsy, laboratory, and diagnostics in the transition to therapy and optimizes interdisciplinary collaboration and decision-making.
The aim of this subproject is to integrate current national and international treatment guidelines and relevant study information via web links in order to support guideline-based therapy decision-making in the tumor board. As a solution, current German and international treatment guidelines will first be integrated into the system. In addition, links to relevant studies and protocols of ongoing studies will be provided. Based on the structured patient data and findings available, a guideline-based first-line therapy recommendation will be generated automatically during the tumor board meeting. This work package only covers the implementation of a first-line therapy recommendation. There are plans to expand the system to include second-line therapy recommendations in the future. The cooperation partners in this project are celsius37, Mannheim University Hospital, and Heidelberg University. In the closed-loop process, the subproject is located between biopsy, laboratory, and diagnostics in the transition to therapy and optimizes interdisciplinary collaboration and decision-making.
The aim of the subproject is to scientifically evaluate the mobile application, the ad hoc tumor board, the asynchronous tumor board, and the decision-making algorithm using prospective studies and existing patient data. The subproject pursues several approaches:
The cooperation partners in this project are celsius37, Mannheim University Medical Center, and Heidelberg University. In the M²OLIE closed-loop process, the subproject is located between biopsy, laboratory, and diagnostics in the transition to therapy and serves to scientifically validate digital decision support before the start of therapy.