

The goal of M²DATA is to develop a Data Lake that functions as a central data infrastructure for the M²OLIE closed-loop. Complex, application-specific data (e.g. data from the biopsy needle or robot movements) will be converted into a standardized data format, linked to patient information from clinical information systems and then made available to researchers and physicians in the intervention room. M²DATA helps to provide patients with important medical information about the planned molecular intervention prior to the treatment, to plan the clinical treatment process efficiently and economically, to provide physicians with information about the current intervention, as well as to enable new medical procedures and findings through a holistic patient image and automated data analysis of already performed interventions. In addition, the Data Lake creates the prerequisites for innovative methods of machine learning to optimize clinical processes (such as the tumor board process) and data analytics to improve medical treatment. In the medium term, M²DATA will serve as the basis for the further development of the M²OLIE research campus into a platform ecosystem. In order to achieve the goals of M²DATA more efficiently, the project was divided into three subprojects: M²HUB, M²OTUS and ProM²etheus.
M²HUB (lead: University of Mannheim) is responsible for the technical implementation of the Data Lake as a central infrastructure and for the scientific research into its potential. Data analytics will be used to optimize the M²OLIE process through innovative indicators and to develop technological innovations. This includes above all the further development of the application started in the last funding phase for the implementation of ad hoc tumor boards and the electronic patient education. In addition, the scientific focus in the second funding phase will be on researching the effects of data-based decision support systems. Especially against the background of the introduction and use of systems with artificial intelligence methods, it is important to find out how medical decision-makers react to semi-autonomous decision-support systems and how the effectiveness of the latter can be maximized. This research helps to derive drivers for acceptance and design guidelines for new applications and to evaluate them within the framework of the implementation of the Data Lake.
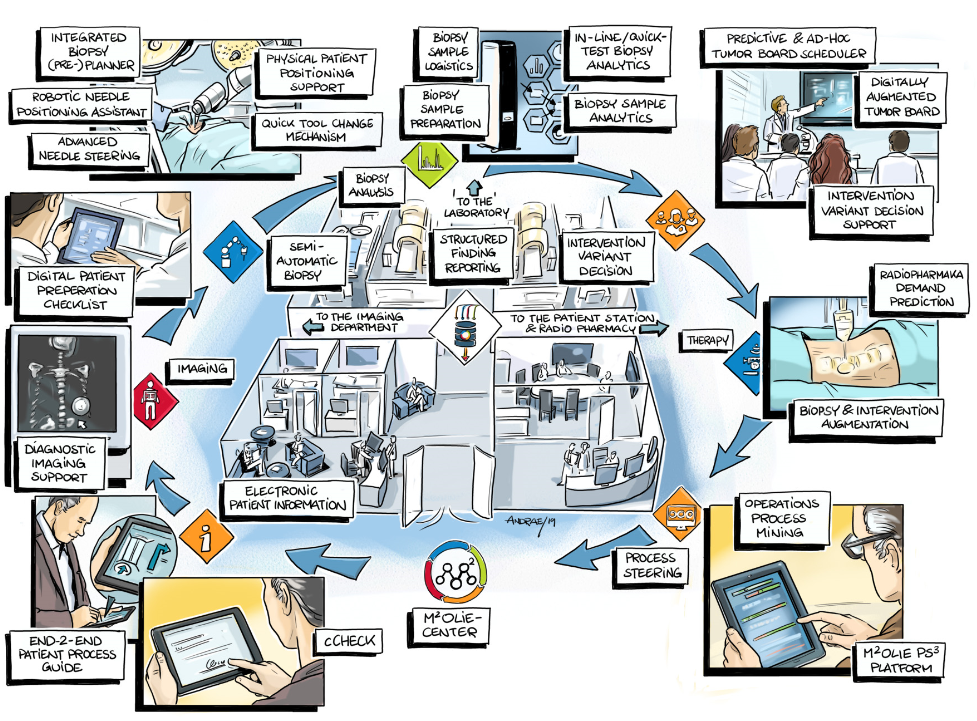




In the subproject M²OTUS (lead: Mint Medical GmbH), the linkage of the process model with the Data Lake takes place. The evaluation of a data component forms the core of an automated tumor board architecture. Tumor boards play a special role within clinical decision-making processes. Due to the systemic character of many cancer diseases, they represent a decision point that brings together clinical data in an interdisciplinary but strictly case-oriented manner.
The pooling of the mostly heterogeneous and only partially structured clinical data in preparation for a tumor board session, however, is still associated with a high manual workload. Tumor boards therefore require an extensive preparation phase in which reports, results and images have to be compiled for decision making. During the tumor board, the case is presented and discussed on the basis of this data base, which is extracted beforehand. Decisions and further steps must be documented and initiated in the further process. Here a direct connection with the Data Lake is vital. Lead times for data preparation are reduced and the tumor board becomes more reactive. Decision making becomes more efficient by ensuring that all decision-relevant data is actually available, thus enabling a faster start of therapy.



The ProM²etheus subproject (lead: Fraunhofer IPA) focuses on the interfaces to the Data Lake on the one hand, but also on the processes and procedures in the intervention room on the other. The first aspect is of fundamental significance for the goals of the joint project, since without (standardized) interfaces a common Data Lake can only be enabled with great difficulty. In addition, standardized interfaces make it easier to integrate other data types that have not yet been taken into account and that may become relevant in the further development of the Research Campus. This is made possible through targeted extensions of existing medical data formats (e.g. DICOM). Furthermore, an interface to the RIS of our partner Mint Medical GmbH is to be created in order to enable close integration of the existing RIS with the processes from M²OLIE (M²OTUS). As part of this measure, the process of the tumor board will also be revised and integrated.
The second focus is on the processes and data from the intervention room. The intervention room has the special feature that it contains many devices that have not yet been used together for interventions. In addition, the intervention room has a modular structure so that it can be flexibly adapted to current requirements at any time. This represents a major challenge for process recording, planning and execution. We do not yet know of any solution that considers this challenge so comprehensively. The integration of the data generated by these devices (e.g. sensor or actuator data) in the DICOM standard format is also new and offers new possibilities, particularly with regard to the use of process knowledge for analysis. For the exploitation of the process manager itself, the prototype developed in the first funding phase is to be further developed into a robust system. This also includes the integration of a process portal, which contains all (sub)processes of the molecular intervention. These processes are to be supplemented with further medical processes outside the molecular intervention, so that the portal finally represents a library of digital medical processes. In order to accelerate and facilitate the process mapping for the portal, an agile process design will be drafted and implemented as a further module.


